suprcat.com
👇 SuPRCat postdoc Amreen Bains in action!

👇 SuPRCat postdoc Amreen Bains in action!
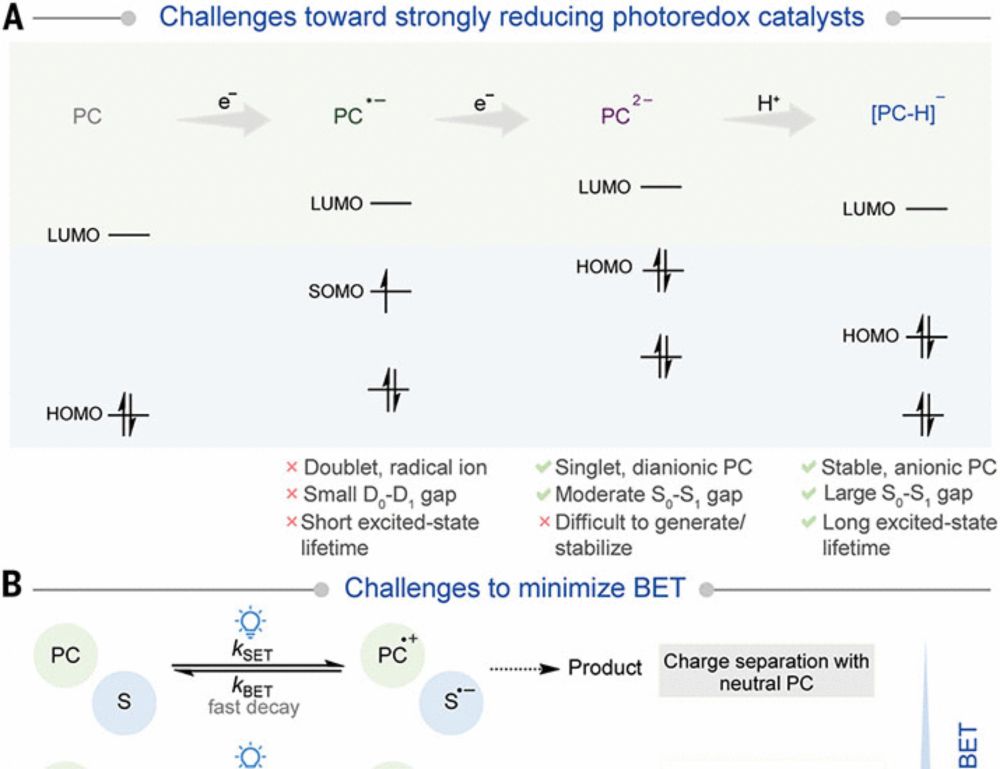
pubs.acs.org/doi/full/10....

pubs.acs.org/doi/full/10....
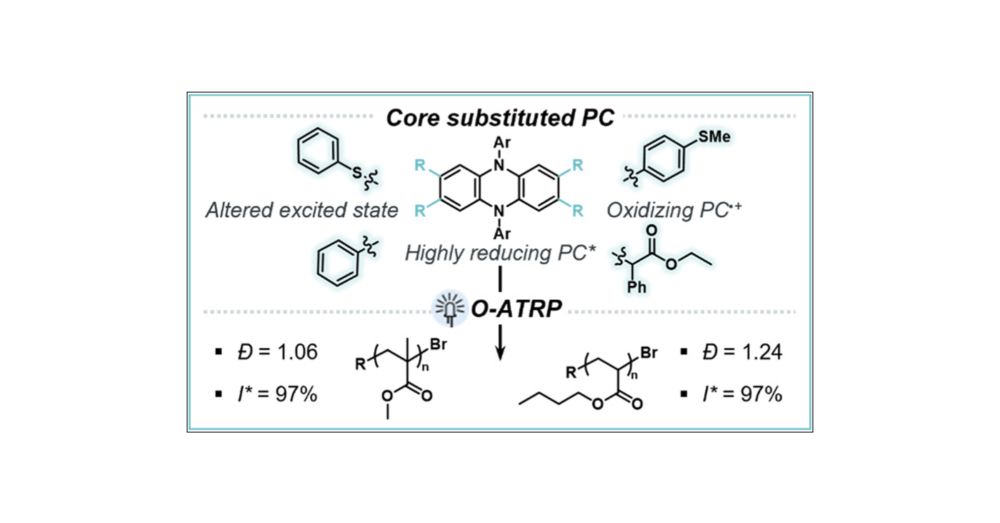

www.chemistryworld.com/news/visible...

www.nature.com/articles/d41...

www.nature.com/articles/d41...


pubs.acs.org/doi/full/10....
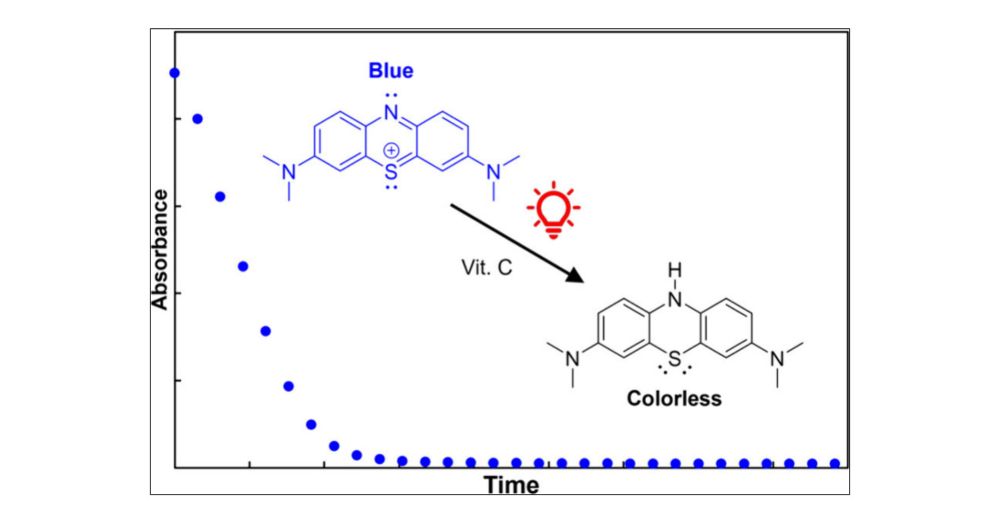
pubs.acs.org/doi/full/10....
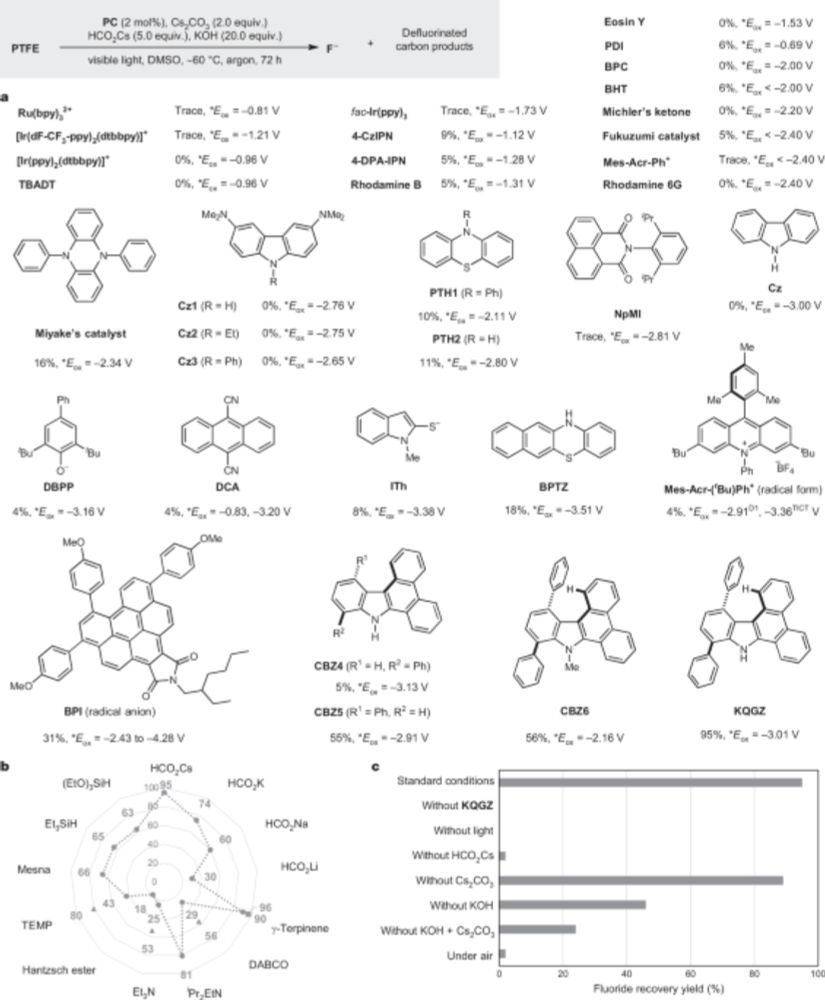
In one, the authors show that powdered Teflon can be transformed into charcoal+fluoride with light+a catalyst+reducing reagent & some very important help from mainly KOH and Cs formate
This is bonkers cool: