Dylan Duerre
@dylanduerre.bsky.social
32 followers
27 following
16 posts
PhD candidate in the Galmozzi Lab at UW-Madison. 🔬 scientist 🧬, 🏔️ wilderness advocate 🌲, 🥅 hockey fan 🏒.
Posts
Media
Videos
Starter Packs
Reposted by Dylan Duerre
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25

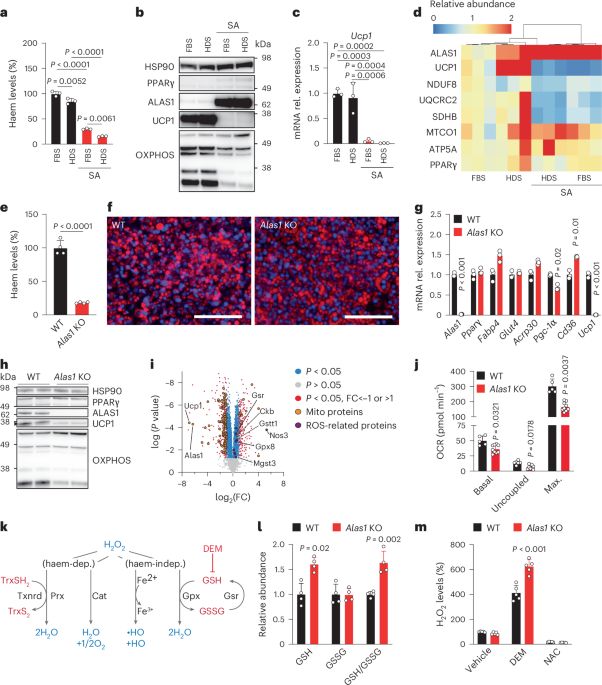
Haem biosynthesis regulates BCAA catabolism and thermogenesis in brown adipose tissue
Nature Metabolism - Inhibition of haem synthesis is shown to lead to the accumulation of branched-chain amino acids in the brown adipose tissue of mice, which reduces UCP1 levels and impairs...
urldefense.com
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25

A BCAA–haem axis regulates brown fat function - Nature Metabolism
Activated brown adipose tissue takes up large amounts of branched-chain amino acids (BCAAs), but their fate remains unclear. We provide evidence of a metabolic link between BCAA catabolism and haem bi...
www.nature.com
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25
Dylan Duerre
@dylanduerre.bsky.social
· Mar 25

Haem biosynthesis regulates BCAA catabolism and thermogenesis in brown adipose tissue
Nature Metabolism - Inhibition of haem synthesis is shown to lead to the accumulation of branched-chain amino acids in the brown adipose tissue of mice, which reduces UCP1 levels and impairs...
urldefense.com