Jeremy Garb
@garbjeremy.bsky.social
46 followers
45 following
15 posts
PhD student at the Sorek lab.
Immune proteins.
Posts
Media
Videos
Starter Packs
Jeremy Garb
@garbjeremy.bsky.social
· Sep 3
Jeremy Garb
@garbjeremy.bsky.social
· Sep 2
Jeremy Garb
@garbjeremy.bsky.social
· Sep 2
Jeremy Garb
@garbjeremy.bsky.social
· Sep 2
Jeremy Garb
@garbjeremy.bsky.social
· Sep 2
Jeremy Garb
@garbjeremy.bsky.social
· Sep 2
Jeremy Garb
@garbjeremy.bsky.social
· Sep 2
Jeremy Garb
@garbjeremy.bsky.social
· Sep 2

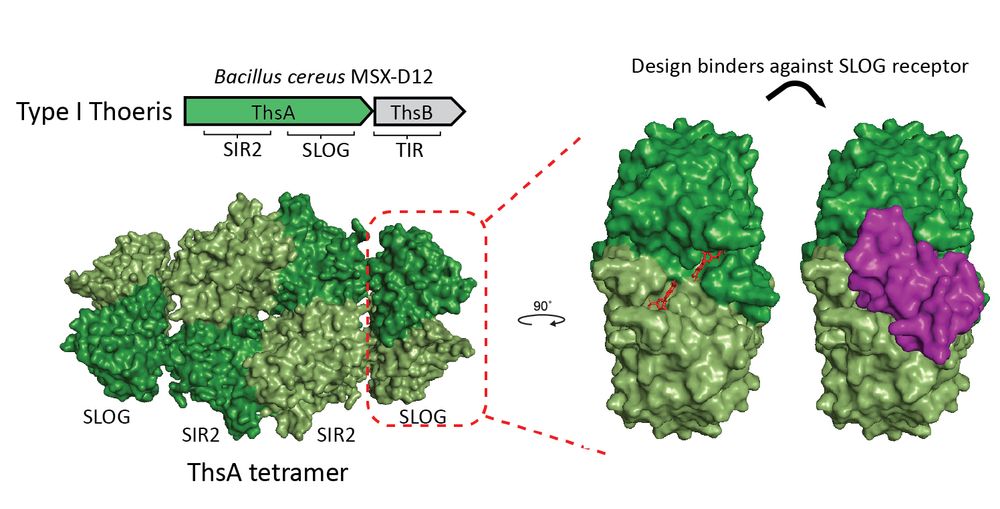
Synthetically designed anti-defense proteins overcome barriers to bacterial transformation and phage infection
Bacterial defense systems present considerable barriers to both phage infection and plasmid transformation. These systems target mobile genetic elements, limiting the efficacy of bacteriophage-based t...
www.biorxiv.org
Jeremy Garb
@garbjeremy.bsky.social
· Nov 16