Axel Delamarre
@axeldelamarre.bsky.social
1.4K followers
1.5K following
19 posts
I study Chromatin structure, organization and function.
Postdoc in the Whitehouse lab, MSKCC, NYC.
Former PhD in the Pasero lab, IGH, France.
Posts
Media
Videos
Starter Packs
Pinned
Axel Delamarre
@axeldelamarre.bsky.social
· Jul 17
Reposted by Axel Delamarre
Reposted by Axel Delamarre
Reposted by Axel Delamarre
Reposted by Axel Delamarre
Mathew Jones
@jonesmjk.bsky.social
· Sep 1

A high-resolution, nanopore-based artificial intelligence assay for DNA replication stress in human cancer cells
Nature Communications - Determining how replication forks move across the human genome is critical for the effective use of agents that target replication stress. Here, the authors present...
rdcu.be
Reposted by Axel Delamarre
Reposted by Axel Delamarre
Reposted by Axel Delamarre
Reposted by Axel Delamarre
Reposted by Axel Delamarre
NealeLab
@labneale.bsky.social
· Aug 10

Condensin and topoisomerases cooperate to relieve topological stress at stalled replication forks
Resolving complex topological structures at replication forks is vital for successful DNA replication, but the mechanisms are little understood. Evidence from diverse eukaryotes suggests that condensi...
biorxiv.org
Reposted by Axel Delamarre
Axel Delamarre
@axeldelamarre.bsky.social
· Jul 17
Axel Delamarre
@axeldelamarre.bsky.social
· May 30

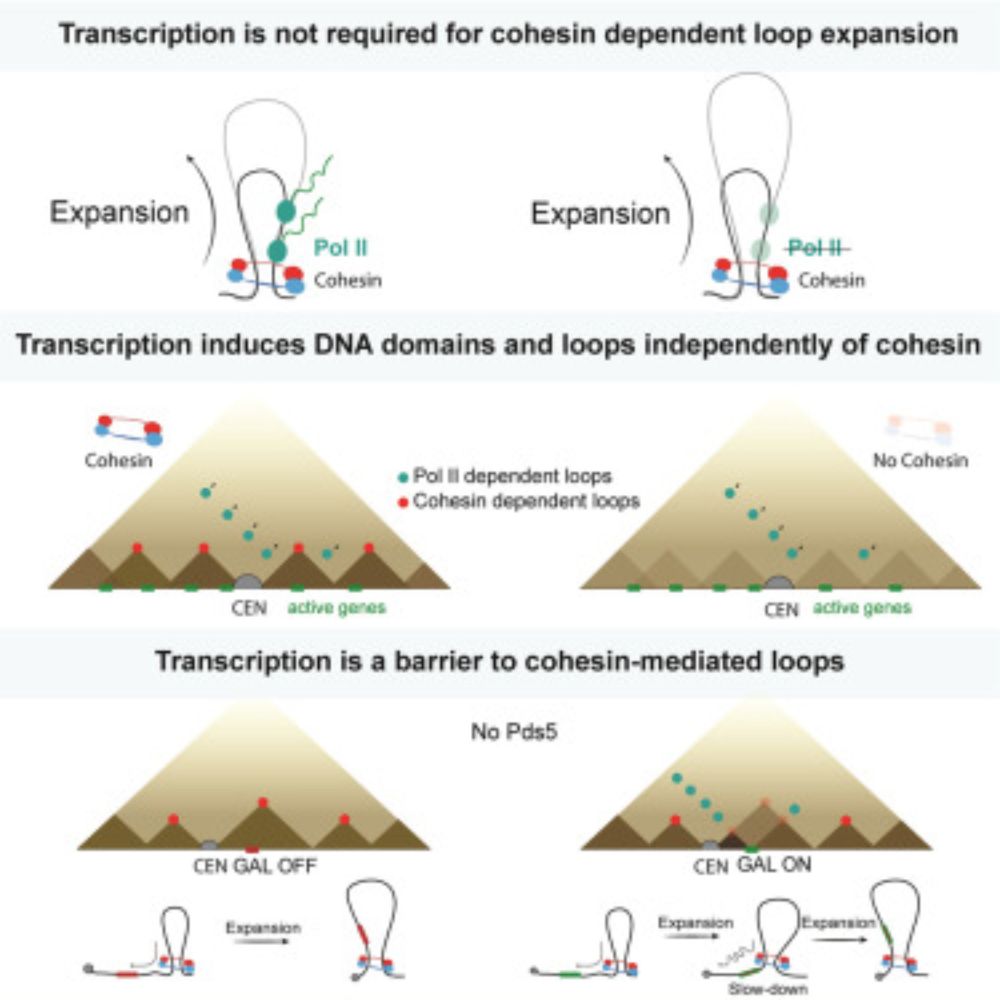
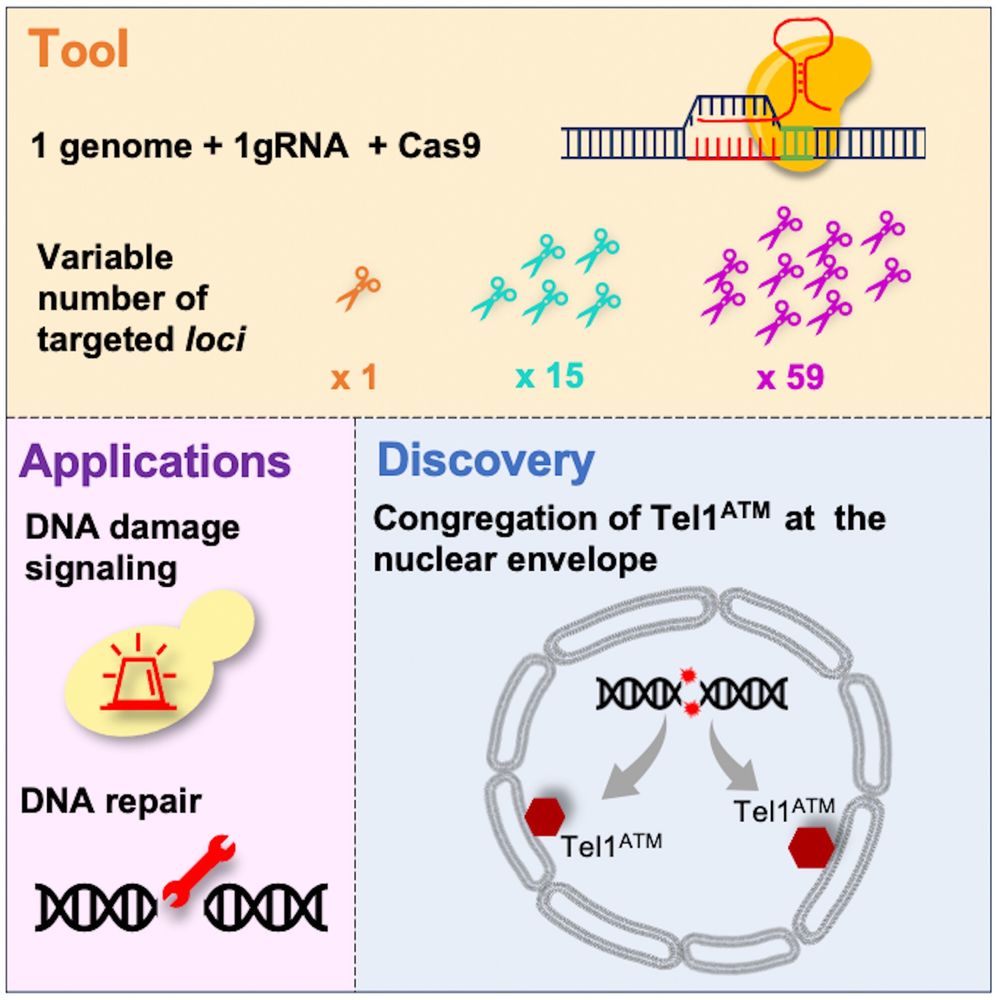
Chromatin architecture mapping by multiplex proximity tagging
Chromatin plays a pivotal role in genome expression, maintenance, and replication. To better understand chromatin organization, we developed a novel proximity-tagging method which assigns unique DNA b...
www.biorxiv.org
Axel Delamarre
@axeldelamarre.bsky.social
· May 30